Maggie Rogers holds in-person box office for upcoming tour “Don’t Forget Me” in Boston
April 26, 2024
BREAKING NEWS: Simmons Class of 2024 Council impeached and stripped of duties
April 25, 2024
VPEO hosts benefit concert for local rape crisis center
April 22, 2024
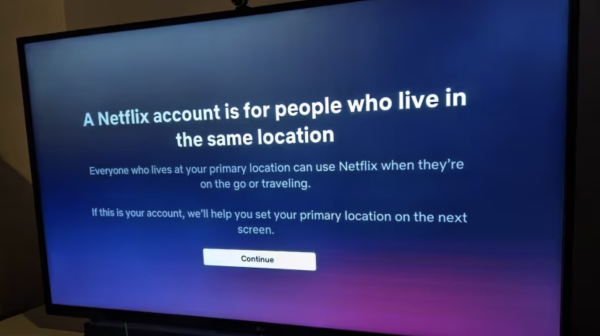
Opinion: Netflix’s household rules are unreasonable
April 16, 2024
SGA creates time capsule to be opened in 50 years
April 15, 2024